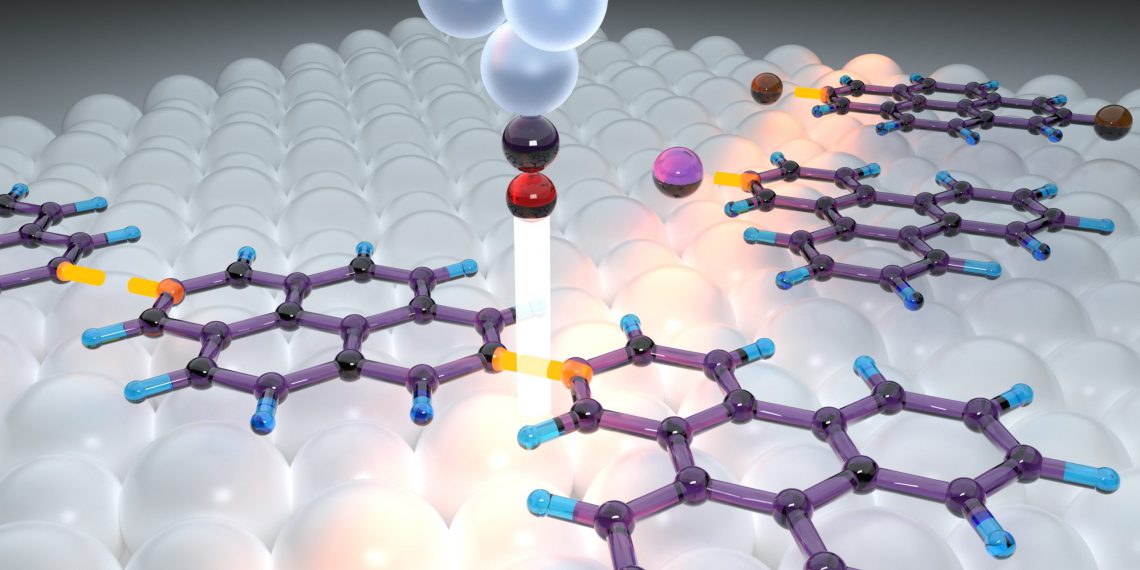
Chemists and physicists at Justus Liebig University Giessen (JLU) have developed a method to assemble organic nanoarchitectures molecule by molecule. To precisely align the molecules in the plane, they use a salt surface on which the molecules can be moved relatively easily. The second tool is the sharp tip of an atomic force microscope, which ends in a single atom or molecule.
This “finger” is used to successively carry out three reaction steps: First, the molecular starting materials are activated to enable a subsequent intermolecular reaction. Then, the previously activated molecular building blocks are pushed together on the surface before the scanning probe tip induces the chemical reaction that leads to the binding of the molecules. These reaction steps are each induced using short voltage pulses applied between the tip and the surface.
The researchers working together in the LOEWE priority PriOSS (principles of surface-supported synthesis strategies) have thus succeeded in answering a question posed by the physicist Richard Feynman as early as 1959 and with which he founded the field of nanotechnology: What would be the impact if we could construct new materials atom by atom? In the early 1990s, engineer K. Eric Drexler formulated the far bolder vision of even having tiny machines perform this task in the future. Such machines would be able to produce almost any nanoarchitecture — assuming chemical stability — and endow them with tailored properties.
Such proposals have been hotly debated for decades. Richard Smalley, winner of the 1996 Nobel Prize in Chemistry, expressed two main counterarguments: First, the individual building blocks (atoms or molecules) could not be aligned precisely enough with the help of the “fingers” (or tools) of such manufacturing machines, since these fingers themselves could not be made infinitely smaller — they would, after all, also consist of atoms. Second, both the molecular building blocks and the products would always stick to the fingers due to adhesion forces. These cogent arguments went down in scientific history as the “fat finger” and the “sticky finger” problems.
The new method circumvents both problems: The inert, i.e., low-reactivity, salt surface takes on the role of a “non-sticky hand,” and the sharp scanning probe tip is the “non-fat finger.” In this way, JLU scientists will be able to produce new organic nanomaterials in the future and systematically investigate how the structure affects their properties. This should make it possible to specifically influence the properties of the nanoarchitectures.
This is particularly interesting for applications in electronic components such as organic field-effect transistors (OFET), light-emitting diodes (OLEDs, e.g. for smartphone displays) or solar cells. In addition, the stepwise induced chemical reactions can provide new insights into the reaction mechanisms of molecules on surfaces.